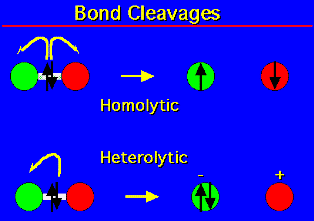
There is no denying the fact that the majority of organic transformations involve the movement of electron pairs indicating heterotypic reactions. There are a few important addition reactions, however, in which the electron reconfiguration involves the movement of single electrons. Whereas heterolytic bond cleavage leads to ion pairs, homolytic bond cleavage results in unpaired electrons – or free radicals. Some weak bonds have a tendency to fragment homolytically (e.g., peroxides, halogens). Chemists use a slight variation of curved arrow notation to show the movement of single electrons. For single electron movement, “fishhook” arrows, i.e., single-headed arrows are drawn.
Bond fission
A covalent bond is formed when electrons are shared between two atoms in the classical sense. A single bond (sigma bond) is thus made up of two electrons. Now a chemical reaction takes place when old bonds are broken and new ones are created.
Homolytic fission
Homolytic fission is where each atom of the bond keeps an electron each resulting in species called free radicals. Radicals are important intermediates in organic chemistry. As the bond breaks to give two similar species each keeping an electron this form of bond breaking is called Hemolytic Fission.
Heterolytic fission
In this case we can see that one of the atoms carries a negative charge after bond cleavage indicating that it has both the electrons of the bond and the other has no electrons at all. Hence it is electron deficient thus positively charged. As the electrons are not divided equally after bond cleavage this is called Heterolytic Fission. In a case the C atom carries a positive charge it is called a carbocation and in the case, it carries both the electrons of the broken bond and is negatively charged, it is quite intuitively called a Carbanion.
We should bear in mind that both types of fissions are applicable to both homoatomic and heteroatomic bonds (bonds between two different atoms say C-N or C-O). Carbocation and Carbanions are the most important carbon intermediates in organic chemistry.